Your One-Stop Resource for COVID-19 Swab Kits & Testing

Puritan: A Leader in Quality COVID-19 Swabs
Puritan has been identified as one of a number of essential suppliers of products needed for COVID-19 testing. As such, manufacturing processes have shifted to meet the contractual obligations to support our government and those essential companies that provide critical testing methods.
We created this Covid-19 Resource Portal for distributors, end-users, and most importantly, healthcare providers to stay up to date on the latest swab and testing updates.
Coronavirus is a type of virus that causes respiratory illness, an infection of the airways and lungs. While COVID-19 is a new strain of coronavirus, it's part of the same family of coronaviruses that includes the common cold.
Here are some of the best resources to learn more about the virus and get up-to-date information:
1. CDC COVID 19 PORTAL
For the most up to date CDC recommendations, protocol, and news, check out the CDC Covid-19 portal here.
2. INFORMATION FOR HEALTHCARE WORKERS
Learn about post-Covid conditions, managing COVID-19 patients, and patient isolation and precautions by clicking here.
3. CLINICAL & PROFESSIONAL RESOURCES
If you're looking for targeted resources for your clinical or professional practice, you'll want to check out the resources in this guide by the CDC here.
Can't find what you're looking for? The Puritan Medical Team is here to help. As a leader in COVID-19 nasal swab testing, we are happy to help you identify the right product or resource to help.
The Types of COVID Swab Tests
For initial diagnostic testing for COVID-19, CDC recommends collecting and testing an upper respiratory specimen. Nasopharyngeal (NP) specimen is the preferred choice for swab-based COVID-19 testing.
When collection of a nasopharyngeal swab is not possible, the following are acceptable alternatives. Here is a full list:
-
Nasopharyngeal (NP) specimen collected by a healthcare professional (HCP)
-
Anterior nares (nasal swab) specimen collected by a HCP or by onsite or home self-collection (using a flocked, a round foam swab, or spun polyester swab)
-
Nasal mid-turbinate swab collected by a HCP or by a supervised onsite self-collection (using a flocked tapered swab)
-
Oropharyngeal (OP) specimen collected by a HCP
-
Nasopharyngeal wash/aspirate or nasal wash/aspirate (NW) specimen collected by a HCP
For more information on substitute swabs and other ways to collect specimens, see current FDA guidelines for COVID-19 testing supplies under the “What If I Don’t Have” section.
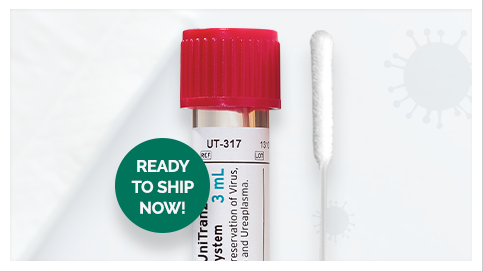
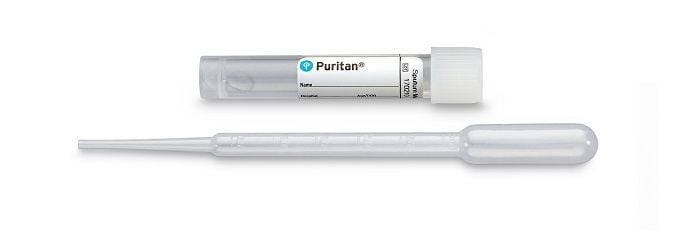
Depending on how the platform used to test the specimen, some tests are designed to analyze a swab specimen directly, without the use of any media. Other platforms require the swabs to be placed immediately into a sterile transport tube containing 2-3mL of either UniTranz-RT® Universal Transport Systems, Opti-Swab® - Liquid Amies Transport Systems, or sterile saline solution.
For those patients with productive coughs, sputum samples might be collected and used for testing Sputum Medium
Puritan's swabs and transport systems have been used in clinics and at-home testing kits since the beginning of the pandemic.
Here are our recommended transport systems and COVID swab products.*
Recommended Puritan Products:
Puritan’s UniTranz-RT® (Universal Transport Systems)
-
Puritan’s UniTranz-RT® is intended for the collection and preservation of viruses (including COVID-19), chlamydia, mycoplasma, and ureaplasma specimens. Our kits ensure the safe transport of clinical specimens when immediate testing is not available. Used in viral antigen detection tests, PCR and rapid tests.
-
UniTranz-RT® meets the CDC’s interim guidelines for collecting, handling and testing clinical COVID-19 specimens from persons of interest. Our flock swabs are the ideal swab for nasopharyngeal and oropharyngeal specimen collection when 2019-nCoV is suspected.
Nasopharyngeal COVID Swabs (NP)

Flock Swabs (NP)
Foam Swabs (NP)
Polyester Swabs (NP)
Nasal COVID Swabs

Foam Swabs (Nasal)
- 25-1506 1PF 100
- 25-1506 1PF SOLID
Polyester Swabs (Nasal)
Flock Swabs (Nasal)
Oropharyngeal (OP) COVID Swabs

Flock Swabs (OP)
Foam Swabs (OP)
- 25-1506 1PF 100
- 25-1506 1PF SOLID
Polyester Swabs (OP)
Additional Flock COVID Swabs:
*All swabs above can also be used in the dry transport tubes variation
*NOTE: All Puritan UniTranz-RT®, Opti-Swab® Liquid Amies collection and transport systems, and Sputum medium are considered final sale, no exceptions. Thank you for your understanding.
Did you know there’s more than one way to take a COVID-19 sample? Knowing more than one method can increase flexibility and help you meet testing demand.
Click Here to Download this FREE new toolkit, to get detailed how-to instructions for taking both nasal and nasopharyngeal samples.
These are particularly helpful because each type of sample collection requires different supplies and different methodology.
This resource is easy to print and share for use in your healthcare clinic, testing site, or at-home testing kit.
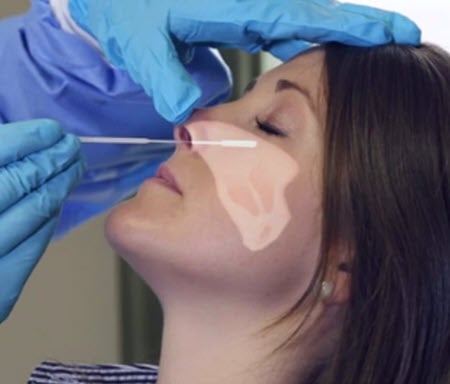
7 Steps for Taking a Nasopharyngeal Swab Specimen
- Wash hands and wear appropriate personal protective equipment.
- Aseptically remove sterile swab from package.
- Tilt patient’s head back slightly to straighten nasal passage.
- Insert swab straight back horizontally to the nasopharynx until resistance is met.
- Rotate swab up to 5 times and hold in place 5-10 seconds to collect sample.
- Place swab into the viral transport medium and break off handle where indicated.
- Cap tightly, label with patient information and deliver to lab for testing.
How to Do a COVID Nasal Swab Test
- Choose a swab with a flexible plastic handle and flock tip such as a 6" Sterile Elongated Flock Swab w/Polystyrene Handle.
- Insert the swab into one nostril straight back (not upwards) and horizontally to the nasopharynx up to the measured distance on the swab handle.
- Rotate the swab up to 5 times and hold in place for 5 to 10 seconds to collect sample material.
- Remove the swab and insert into a vial containing 1 to 3 ml of viral transport media.
- Break the swab handle at the scored breakpoint line.
- Label the vial with appropriate patient information.
COVID-19 Questions
Need additional information on Puritan COVID-19 products?
See our Specimen Collection and Diagnostic Products >
Please Note: Because news about COVID-19 is particularly fluid, we will update this portal as information becomes available.

%20(1).png?width=500&name=Collect%20COVID-19%20Specimens%20(2)%20(1).png)

